Learn more about Fluids, Electrolytes, Acids & Bases
In this module, you will learn more about
- The body fluid compartments
- Water balance
- The role of electrolytes
- Regulation of water and electrolyte balance
- Acid and base balance
Learn even more: see Chemistry, Cells, Respiratory system and Urinary system modules
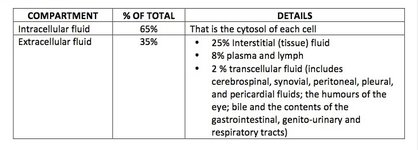
1 The body fluid compartments
Body fluids are then water and whatever is dissolved in the water which includes: salts and other low molecular weight molecules and proteins. The body fluid is distributed between different fluid compartments. Each compartment is bounded by one or more layers of cells. There is constant exchange of water between the compartments.
Osmolarity
When describing the properties of a solution, a distinction is made between osmolarity and concentration:
For example the concentration of sodium in the plasma is around 150 millimoles per litre (150 mM), whereas the plasma osmolarity is a different value (around 300 milli-osmoles per litre, 300 mOsm/L) because it has to, in addition to sodium, account for the amounts other dissolved ions (particularly chloride), proteins, glucose etc.
By the way:
biochemistry labs in health facilities will report osmolality not osmolarity and normal range for the plasma osmolarity is 275 to 295 milli-osmoles per kilogram of plasma
- the osmolarity is the TOTAL amount of molecules and ions dissolved in a solution
- concentration relates to the amount of one particular molecule or ion in a solution
For example the concentration of sodium in the plasma is around 150 millimoles per litre (150 mM), whereas the plasma osmolarity is a different value (around 300 milli-osmoles per litre, 300 mOsm/L) because it has to, in addition to sodium, account for the amounts other dissolved ions (particularly chloride), proteins, glucose etc.
By the way:
biochemistry labs in health facilities will report osmolality not osmolarity and normal range for the plasma osmolarity is 275 to 295 milli-osmoles per kilogram of plasma
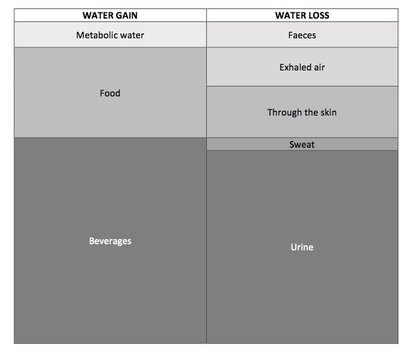
2 Water balance
The amount of water lost must be balanced by water intake/production. An imbalance in either direction has adverse health outcomes.
A few comments on the details of the table above:
A few comments on the details of the table above:
- The size of the section reflects the relative contribution to either water gain or water loss
- “Metabolic water” is the water produced as a result of aerobic respiration in mitochondria
- Water loss through sweat can vary with body temperature and sweat also accounts for loss of body salts
- The principal ways the body achieves water balance is through: (i) regulation of water gain through eating and drinking; and (ii) regulation of water loss through adjusting urine output
Movement between the fluid compartments
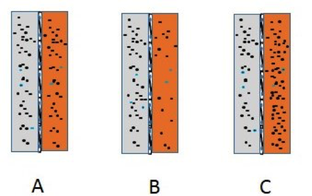
Just as movement of water in-and-out of cells is driven by osmosis, so too is the movement of fluid between compartments.
Consider the three situations above where there are two fluid components (say interstitial fluid is the grey compartment and the orange compartment represents the plasma)
In both B and C there can be a net movement of water between the compartments to compensate for this and restore the balance towards an equality of the osmolarity of both compartments.
Consider the three situations above where there are two fluid components (say interstitial fluid is the grey compartment and the orange compartment represents the plasma)
- In A the osmolarity of both compartments is equal
- In the B there is an imbalance in favour of the interstitial fluid
- In C there is an imbalance in favour of the plasma
In both B and C there can be a net movement of water between the compartments to compensate for this and restore the balance towards an equality of the osmolarity of both compartments.
3 The role of electrolytes
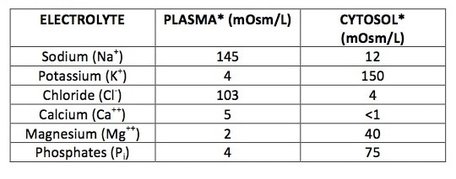
Any substance when dissolved in water that is capable of carrying a charge is an electrolyte. These are the cations and anions of solutions. In a clinical setting the same terminology is used although generally when electrolyte balance is considered this is restricted to considering the plasma concentrations of sodium, potassium, chloride and bicarbonate (HCO3-)
Calcium
Calcium has a major role in structural support in the matrix of bones. Increases in cytosolic calcium is a critical event in the release of neurotransmitters from neurons, contraction of muscle fibres, and many chemical reactions which occur inside cells. Calcium is an essential requirement for blood clotting as well.
Chlorine (as chloride ions)
This is the principal anion of the extracellular fluids (as illustrated by plasma in the table above). Chloride and bicarbonate exchange between specific fluid compartments serves an important function in several physiological processes.
Iron
Iron has several different charge states including Fe++ and Fe+++. In the body its principal role is as a component of enzymes and proteins the roles of which include: oxygen transport, nucleotide synthesis and fighting infections.
Phosphates
This is actually a collection of anions (H2PO4-, HPO4--, and PO4---). These have a central role in the synthesis of ATP, phospholipids and in regulating the function of many enzymes. There are a major component of mineral matrix of bones. In body fluids they also have a role as buffers (described in a separate section)
Potassium
This is the principal cation of the cytosol (as illustrated in the table above). Generation of the action potential is also dependent on maintaining a higher concentration of potassium in the cytosol compared to that of the body fluids. For example, a substantial increase in plasma potassium levels can lead to cardiac arrhythmia. Another important role of potassium relates to its transport across fluid compartments where potassium transport is linked with the transport of other molecules (e.g. sodium and hydrogen ions).
Sodium
This is the principal cation of the extracellular fluids (as illustrated by plasma in the table above). Indeed sodium, along with its associated anions, including chloride, account for more the 90% of the total plasma osmolarity. As a result of this, sodium plays a central role in maintenance of water balance. For example, a dramatic loss of plasma sodium would lead to major water shifts into the tissues leading to hypotension and tissue oedema. Generation of the action potential is also dependent on maintaining a higher concentration of sodium in the body fluids compared to that of the cytosol.
Another important role of sodium relates to its transport across fluid compartments where sodium transport is linked with the transport of other molecules (e.g. amino acids, monosaccharides and hydrogen ions).
Another important role of sodium relates to its transport across fluid compartments where sodium transport is linked with the transport of other molecules (e.g. amino acids, monosaccharides and hydrogen ions).
4 Regulation of water and electrolyte balance
Calcium and phosphates
The regulation of plasma calcium and phosphate levels are linked.
Calcitriol is a highly potent form of vitamin D which will:
A drop in blood calcium levels triggers an increase in blood parathyroid hormone levels which:
Blood phosphate levels are prevented from becoming too high by an overflow mechanism. That is, there is a limit to how much phosphate can be reabsorbed by the kidneys, any excess is lost in urine.
Also phosphate reabsorption can be inhibited by parathyroid hormone
Calcitriol is a highly potent form of vitamin D which will:
- increase bone resorption and in this way increase plasma levels of both calcium and phosphates; and
- increase calcium absorption in the gastro-intestinal system.
A drop in blood calcium levels triggers an increase in blood parathyroid hormone levels which:
- increases synthesis of calcitriol;
- increases bone resorption; and
- increases calcium reabsorption in the kidneys
Blood phosphate levels are prevented from becoming too high by an overflow mechanism. That is, there is a limit to how much phosphate can be reabsorbed by the kidneys, any excess is lost in urine.
Also phosphate reabsorption can be inhibited by parathyroid hormone
Chlorine (as chloride)
Chloride as the major anion of extracellular body fluids essentially tracks the movement of sodium between the fluid compartments. However as mentioned earlier there are several physiological process which involve exchange of chloride and bicarbonate between specific fluid compartments.
Iron
Unlike other electrolytes, there is no mechanism which regulates excretion of iron. Excess iron in the body fluids is readily stored in cells. Increased iron demand can increased export of iron into the blood. Iron release from storage into the blood can be inhibited by a liver-derived hormone, hepcidin.
Potassium
The principal way that plasma potassium level is regulated is by removing excess potassium. There are two principal mechanisms involved here:
- Increased local increases in plasma potassium trigger increased potassium secretion in the renal tubules
- Increased plasma potassium is a major trigger for aldosterone release. In this case potassium secretion is linked with sodium reabsorption
Water and sodium
Given that changes in plasma sodium level will effect overall plasma osmolarity and the movement of fluid between body components: water and sodium balance are linked.
Water and sodium balance are best thought of as regulation of the plasma osmolarity and plasma volume. More particularly:
Water and sodium balance are best thought of as regulation of the plasma osmolarity and plasma volume. More particularly:
- an increase in plasma osmolarity triggers thirst and leads to a reduction in water loss from the body
- a decrease in plasma volume (and thus blood pressure) triggers reduced loss of sodium and water from the body
This one may need a bit more explanation. There is a reflex that prevents the right atrium from being overfilled with blood, as that could lead to impaired contraction. If too much blood is reaching the right atrium via the superior and inferior vena cavae (that’s the plural of cava), then the atrium and vena cavae are stretched and signals are sent to the brain to inhibit ADH release. This means more water is lost in the urine and blood volume drops so the atrium won’t be overstretched. In the flowchart, when blood volume/pressure drops => this reflex is turned off and less water is lost in the urine and blood volume increases.
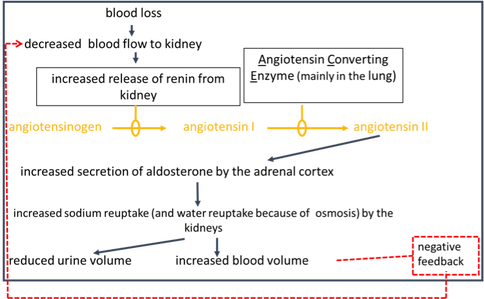
What this flow chart is illustrating, is one way in which the Renin-Angiotensin-Aldosterone-System (RAAS) works.
A drop in blood pressure triggers the release of an enzyme, renin, into the blood stream. Renin will convert an inactive plasma protein, angiotensin to angiotensin I.
Another enzyme, Angiotensin-Converting Enzyme (ACE), will convert angiotensin I to angiotensin II. Angiotensin II is a major trigger for aldosterone release.
Sodium reuptake (reabsorption) is coupled with potassium secretion. The osmotic imbalance that occurs drives water movement by osmosis.
In all three diagrams you can see these responses can be turned off (“negative feedback”) when:
A drop in blood pressure triggers the release of an enzyme, renin, into the blood stream. Renin will convert an inactive plasma protein, angiotensin to angiotensin I.
Another enzyme, Angiotensin-Converting Enzyme (ACE), will convert angiotensin I to angiotensin II. Angiotensin II is a major trigger for aldosterone release.
Sodium reuptake (reabsorption) is coupled with potassium secretion. The osmotic imbalance that occurs drives water movement by osmosis.
In all three diagrams you can see these responses can be turned off (“negative feedback”) when:
- plasma osmolarity is too low (e.g. over-hydration); or
- blood pressure is too high.
5 Acids and acid bases
As part of normal metabolism, hydrogen ions (H+) are produced the accumulation of which would lead to a fall in body fluid pH.
Stomach acid production leads to a release of bicarbonate and, if this was to accumulate, the body fluid pH would rise.
There are three components to how the body deals with these changes in pH in the body fluids
Stomach acid production leads to a release of bicarbonate and, if this was to accumulate, the body fluid pH would rise.
There are three components to how the body deals with these changes in pH in the body fluids
- Buffers in the body fluids
- Respiratory compensation
- Renal compensation
Buffers
The action of buffers is explained in the Chemistry module. The important buffers in the body fluids are
The carbonic acid/bicarbonate buffer pair
CO2 + H2O <=>H2CO3<=>H+ + HCO3-
CO2 + H2O <==H2CO3<==H+ + HCO3-
Considering the equations above, the way this buffer pair works is best conceived where this is a fall in pH. This would lead to an increase in hydrogen ions which are essentially “mopped up” by bicarbonate. The resultant carbonic acid readily decomposes to carbon dioxide and water and in this way the excess hydrogen ions can be considered to be “locked up” in water.
As to what happens with carbon dioxide….
- The carbonic acid/bicarbonate buffer pair
- Proteins
- Phosphates
The carbonic acid/bicarbonate buffer pair
CO2 + H2O <=>H2CO3<=>H+ + HCO3-
CO2 + H2O <==H2CO3<==H+ + HCO3-
Considering the equations above, the way this buffer pair works is best conceived where this is a fall in pH. This would lead to an increase in hydrogen ions which are essentially “mopped up” by bicarbonate. The resultant carbonic acid readily decomposes to carbon dioxide and water and in this way the excess hydrogen ions can be considered to be “locked up” in water.
As to what happens with carbon dioxide….
Respiratory compensation
Elevation of blood carbon dioxide level (pCO2) is the trigger for an increase in respiration.
So now you can see what happens to the carbon dioxide. It is prevented from accumulating in blood by being exhaled in the lungs.
Conversely if blood pH is too high, respiration is decreased, carbon dioxide accumulates lead to an accumulation of bicarbonate.
So now you can see what happens to the carbon dioxide. It is prevented from accumulating in blood by being exhaled in the lungs.
Conversely if blood pH is too high, respiration is decreased, carbon dioxide accumulates lead to an accumulation of bicarbonate.
Renal compensation
There are linked ways in which the kidneys regulate plasma pH:
The amount of these that take place is determined by the pH of the plasma that courses through the nephrons.
The link between the two ways mentioned is brought about by the set of reactions of the carbonic acid/bicarbonate buffer pair. An enzyme carbonic anhydrase can promote the inter-conversions between carbon dioxide & water and bicarbonate & hydrogen ions and it contributes to effective renal compensation.
- Regulating the secretion of hydrogen ions
- Regeneration and reabsorption of bicarbonate
The amount of these that take place is determined by the pH of the plasma that courses through the nephrons.
The link between the two ways mentioned is brought about by the set of reactions of the carbonic acid/bicarbonate buffer pair. An enzyme carbonic anhydrase can promote the inter-conversions between carbon dioxide & water and bicarbonate & hydrogen ions and it contributes to effective renal compensation.
Bibliography
Saladin K Anatomy & Physiology 7th edition, McGraw Hill Education
Osmolality-the blood test, Medline Plus, US National Library of Health, https://medlineplus.gov/ency/article/003463.htm (accessed 6/11/2016)
Osmolality-the blood test, Medline Plus, US National Library of Health, https://medlineplus.gov/ency/article/003463.htm (accessed 6/11/2016)