Learn more about Chemistry
In this module, you will learn more about
- Atoms
- Elements
- Molecules
- Compounds
- Mixtures
- Chemical reactions
- Solutions
- Ions and electrolytes
- Acids and bases
- The pH scale
- Buffers
- Three major classes of macromolecules: carbohydrates, proteins and lipids
1 Atoms
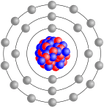
All the things in the world (and actually the whole universe) are made of atoms.
In the simplified diagram, you can see an atom is made up of three types of particles:
- neutrons and protons which are clustered in the centre to form the nucleus
- electrons which orbit the nucleus
Atoms will vary in the number of neutrons and protons in the nucleus and the corresponding number of orbiting electrons.
2 Elements
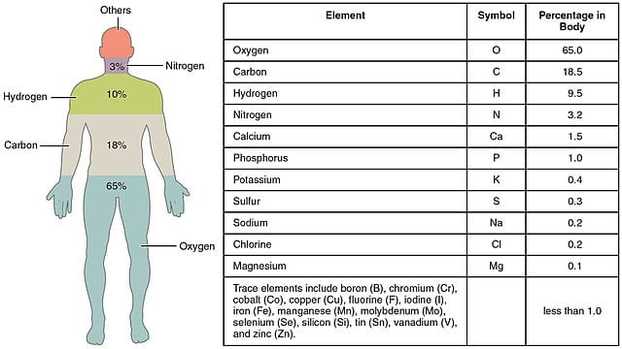
In the table, you can see the most common elements found in the body. There are a couple of important 'take-home' messages here.
- Each element has a symbol made up of one to two letters
- That symbol does not always include the first letter in the name of the element. There are historical reasons for this.
These symbols are used on bags of fluids given to patients, clinical notes and blood tests so it is a good idea to memorise them.
3 Molecules
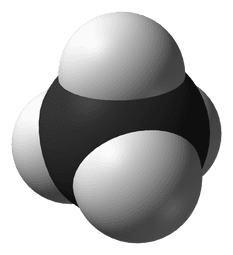
In the diagram, you can see an example of a molecule of oxygen. A few things to note:
- This representation looks a lot different to the earlier diagram of an atom. It is called a space-filling diagram and it is a bit closer to how atoms and molecules are in reality where each ball is an atom and the surface of the ball represents the region where the outer electrons orbit
- In the body, oxygen is found as a molecule of two atoms
Atoms in a molecule are described as being bonded together. The bond comes about by sharing of the outermost electrons between adjacent atoms in molecule. This makes for a very strong bond.
4 Compounds
Each molecule can be represented by a chemical formula that indicates which atoms are present and how many of each type of atom are present.
The formula is a simple way to describe a molecule but it does not give any information about how the atoms in a molecule are arranged. This can be important as there are some molecules (isomers) with the same combination of atoms but in each molecule they are arranged differently.
5 Mixtures
For example, saline for irrigation is a mixture of two compounds: sodium chloride and water
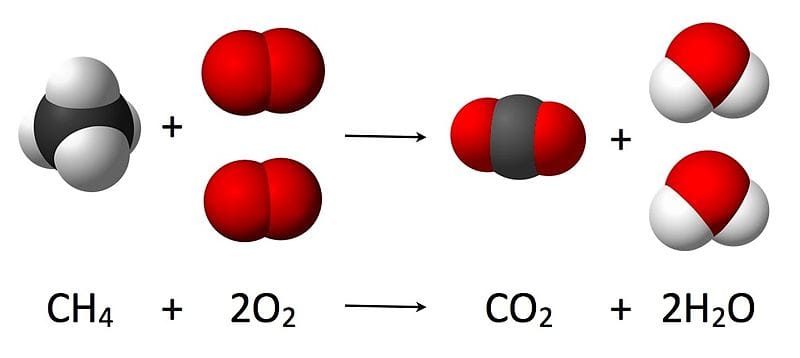
6 Chemical reactions
The starting materials are the ‘reactants’. The end results of the reaction are called the ‘products’. The amount of each reactant determines how much product will be made by the reaction.
In the real world, we do not work by adding single atoms to other single atoms since we cannot separate them at that level. Instead we use a measure termed a ‘mole’.
One mole of any chemical substance contains the same number of atoms or molecules and it is expressed in grams (g).
A mole contains 602,000,000,000,000,000,000,000 atoms or molecules of the chemical of interest.
7 Solutions
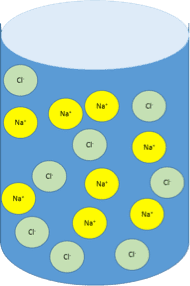
In a solution of salt water, like the photo, the salt is the solute, the water is the solvent.
In the human body, the fluid inside your cells are solutions and water is the solvent.
When describing how concentrated a solution is, we use what is called ‘molarity’ (the symbol is ‘M’). It tells us how much solute is dissolved in a solution. For example:
- a 1M solution contains 1 mole of atoms or molecules in one litre of fluid
- a 2.5M solution contains 2.5 times one mole of atoms or molecules in one litre of fluid
- a 0.5M solution contains half of one mole of atoms or molecules in a one litre solution
- the molarity of normal saline is 150 millimolar (mM) or one-hundred-and-fifty thousandths (0.150) of a mole of NaCl per litre of water which works out at about 9 g of NaCl
Molarity is the concentration measurement used on pathology reports to indicate the levels of electrolytes in the plasma or other molecules in the body for diagnostic purposes.
However, molarity is not the only way we can describe the concentration of a solution. We can also describe it as %w/v which is how many grams of solute is dissolved in 100 mL of solvent. So that 150 mM NaCl could also be expressed as 0.9% w/v.
8 Ions and electrolytes
In any atom, the protons each have a single positive charge and each electron has a corresponding negative charge. In a neutral atom: the number of electrons = the number of protons.
When some compounds (for example sodium chloride/table salt/NaCl) are dissolved in water, the atoms become separated but the outermost electrons don’t get evenly shared. Sticking with the example of NaCl:In some cases, atoms in a molecule don’t separate into individual atoms but instead stay as a molecular ion. Here are some examples:
When ions are present in water, then the water can conduct an electrical current. For this reason, the source of the ions is called an electrolyte. In clinical practice electrolytes are used to describe the ions dissolved in body fluids.
Not all compounds will dissociate into ions when in solution. Sugars such as glucose are an example of this in that they readily dissolve in water but the molecules of glucose don’t ionise.
9 Acids and bases
In solution, a base is an acceptor of hydrogen ions. Here is a generic chemical reaction to illustrate this in which 'HX' is the acid and 'X' is corresponding base:
10 The pH scale
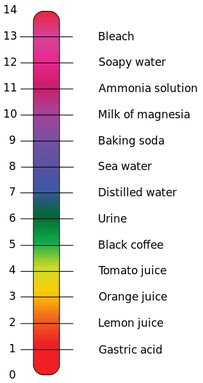
It tells us whether a solution is acidic, alkaline (basic) or neutral.
The pH scale ranges from 0 to 14 with the central point of 7 being neutral. Acidic solutions have pH values below 7 and alkaline solutions have values above 7.
In pure tap water there are 0.0000001 moles of hydrogen ions per litre of water. This method of writing out the hydrogen ion concentration is cumbersome. The pH scale is a simpler method to describe the hydrogen ion concentration. It utilizes the number of decimal places: 0.0000001 becomes pH 7. That is, the decimal place is seven places to the left of the numeral ‘1’.
Another example, which is an acid, is gastric juice. It has a concentration of hydrogen ions equal to 0.01M. The pH is then 2.
An alkaline solution in the body is pancreatic juice. It has a hydrogen ion concentration of 0.00000001M. The pH of pancreatic juice is therefore 8. (For those old enough to remember learning how to use logarithmic tables in school, the calculation for this is based in log to the base ten (log10) mathematics).
It is important to note that small changes in pH are reflected in large changes of hydrogen ion concentration.
The concentration of hydrogen ions in body solutions can have a profound effect on body functions. Normal pH ranges from 7.35 to 7.45 for arterial blood.
11 Buffers
Buffers are made up of a pair of molecules where one is a base and the other an acid.
If the pH is too high (indicating it is too alkaline) then the acid dissociates to release hydrogen ions returning the pH to normal.
If the pH is too low (indicating it is too acidic) then the base can mop up the excess hydrogen ions.
An important example in the body is the carbonic acid – bicarbonate buffer system.
The definition of an acid is that is donates its hydrogen. Carbonic acid (H2CO3) releases one hydrogen to form a bicarbonate ion and a hydrogen ion.
The definition of a base is that it will accept a hydrogen ion. Bicarbonate (HCO3-) ion can pick up a free hydrogen ion to form carbonic acid.
12 Three major classes of macromolecules: carbohydrates, proteins and lipids
A lot of substance in your body is made up of macromolecules.
Three major classes of macromolecules significant to the human body are:
- Carbohydrates
- Proteins
- Lipids
In all three, carbon is a major component. Thus these three classes of macromolecules are part of the bigger group of organic compounds.
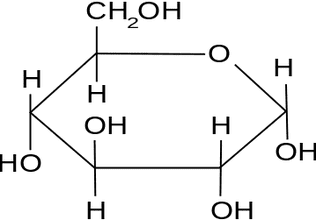
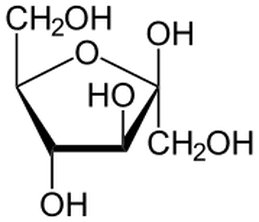
The simplest carbohydrates are monosaccharides ('single sugars'). These are single chains of carbon with hydrogen and –OH groups attached to the carbons. In solution the longer chains will form rings.
In the diagrams, you see two common monosaccharides —glucose and fructose — shown in their ring forms.
Count how many of each atom is present. (The artists have used a common short hand where each 'corner' of the ring is where a carbon atom actually is).
Fructose and glucose are isomers of each other and the body treats each one differently.
Join two monosaccharides together and that is a disaccharide. Sucrose (table sugar) is a disaccharide of glucose and fructose. Lactose is a disaccharide of glucose and galactose.
Join many, many, many monosaccharides together and that is a polysaccharide. Animals store glucose in the form of the polysaccharide glycogen. Starches are plant polysaccharides made from glucose.
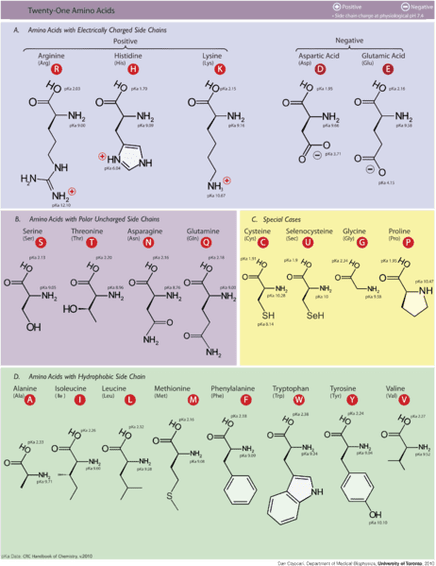
Each protein is a unique chain of many, many amino acids.
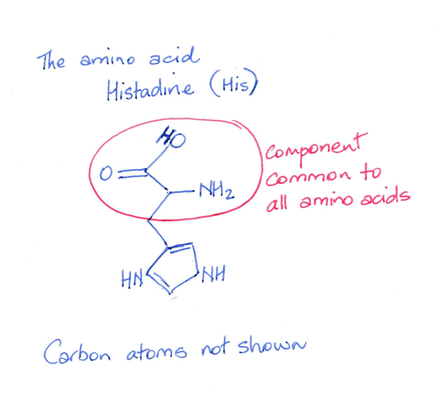
Each amino acid is an organic molecule and it will have one section in common with all other amino acids and one section that is unique – a side chain. The side chain gives it specific chemical properties.
The properties of the protein are determined by which amino acids are included and their sequence in the chain. The side chains on the amino acids in the protein determine the protein’s shape, it’s resistance to being stretched and it’s capacity to bind with other molecules. In this way, a protein’s structure and function are linked.
The local environment of a protein are important for its function. Temperature, pH, and the number of ions present, must be correct for correct function. Changes in the local environment can limit function and severe cases, such as acid environments or very high fevers, can damage proteins irreparably altering the protein and leaving it non-functional (termed ‘denatured’ which means unable to do what nature intended).
Proteins: the special case of enzymes
One group of proteins, enzymes, are biological catalysts. A catalyst is an agent that may either be essential for a chemical reaction to take place or will greatly increase how fast a chemical reaction will take place.
Most of the chemical reactions that keep the body alive and function would be too slow if it wasn’t for the presence of an enzyme.
Proteins are a unique combination of amino acids. This means that each protein has a unique three-dimensional structure.
Just like each house will contain uniquely structured individual rooms, so each protein will have parts (called 'sites' or 'domains') whose three-dimensional structures are unique to that protein.
In the case of enzymes this means that each enzyme will possess an active site which has a unique three-dimensional structure that can only accommodate particular molecules (or maybe a particular part of a macromolecule).
So for each chemical reaction in the body there will be a unique enzyme. For example:
- The enzyme which splits lactose into glucose and galactose is a completely different protein to one that chops up starches into glucose molecules. That is why we can all digest starch but some of us lose the ability to breakdown lactose.
- In the body, we have a specific enzyme that can release the glucose molecules from starch, which is a particular type of carbohydrate. However we do not have an enzyme that can break down cellulose which is the carbohydrate we find in plants.
Like carbohydrates, lipids are macromolecules largely made from carbon, hydrogen and oxygen. The major difference is that lipids have less oxygen present and the arrangement of the atoms means that lipids are not readily dissolved in water.
There are four major types:
- fatty acids
- triglycerides
- phospholipids
- sterols
Examples are shown in the diagrams, and you will see:
- They both have the same end group.
- Like with the drawing of amino acids, none of the carbons are drawn in. They are to be found in between the lines. which represent bonds between adjacent carbon atoms
- The hydrogen atoms that would be bonded to the carbon atoms aren’t drawn in either.
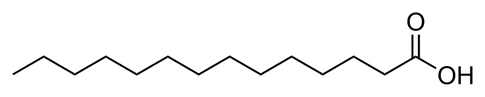
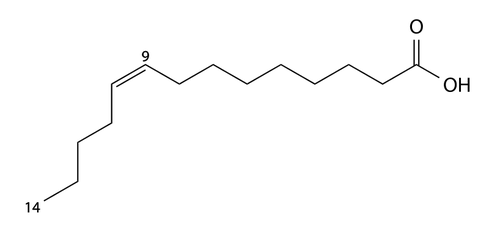
Each fatty acid will have a particular number of carbons in its chain.
The bond between two atoms in molecule involves sharing electrons.
Sharing a single pair is called a single bond represented by a single line. The upper molecule only has single bonds. Chemists categorise this as a saturated fatty acid.
Sharing two pairs is called double bond represented by a double line. The lower molecule has both single and double bonds and so is categorised as an unsaturated fatty acid
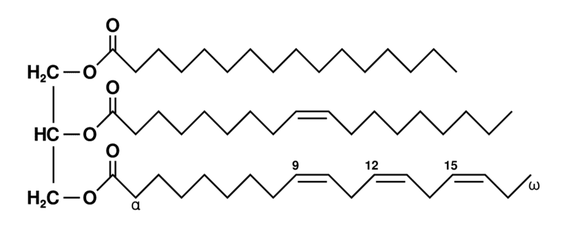
These are the most abundant lipids in the human body and in food.
Which ones are joined will vary from molecule to molecule.
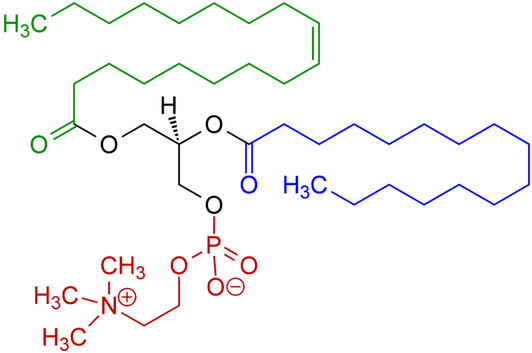
However, when you look at the diagram, you see there are some clear differences:
- There are only two fatty acid chains
- The third binding site for glycerol is not occupied by a fatty acid but instead by a phosphate-containing chemical group. Different phospholipids will vary in the chemical composition of this part except in every case there will be a phosphorus atom
The difference in structure means that unlike triglycerides (which won’t interact with water) phospholipids have a part of each molecule (the part with phosphorus in it) that does interact with water.
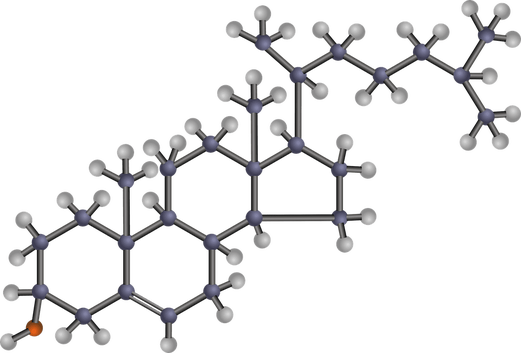
Aside from its own functions, cholesterol is the source of many different sterols found in the body.
In this diagram of a sterol, all the atoms are drawn in: the ring of carbon, the hydrogen and a single oxygen atom.
Like triglycerides, cholesterol does not interact well with water.