Learn more about the the Cardiovascular system
In this module, you will learn more about
- The role of plasma and the cellular components of blood
- The structure of the heart
- Heart function: pacemaker cells and the conduction of electrical signals
- The cardiac muscle and how it contracts
- The normal ECG and what it represents
- The structure of different types of blood vessels
- Cardiovascular haemodynamics
Learn even more: see Homeostasis, Musculoskeletal system, Nervous system, Respiratory system and Immune system modules
1 The role of plasma and the cellular components of blood
Blood and its function
Blood is a connective tissue composed of formed elements and a fluid matrix known as plasma. It carries nutrients and oxygen (O2) to all cells of the body and removes waste products and carbon dioxide (CO2) from these cells.
Blood is a viscous fluid with a pH 7.35–7.45 making it slightly alkaline (basic).
8% of our body weight is blood with men having approximately 5–6 L and women 4-5 L.
Functions of blood:
Blood is a viscous fluid with a pH 7.35–7.45 making it slightly alkaline (basic).
8% of our body weight is blood with men having approximately 5–6 L and women 4-5 L.
Functions of blood:
- Delivers O2 from the lungs and nutrients from the digestive system to all cells within the body.
- Removes metabolic waste and CO2 from all regions of the body. Metabolic waste is excreted via the kidneys and CO2 via the lungs.
- Transports hormones.
- Regulates body temperature.
- Maintains the pH throughout all tissues; proteins in the blood act as buffers.
- Prevents blood loss. Platelets and plasma proteins promote blood clotting.
- Prevents infection. Blood contains antibodies, complement proteins and white blood cells (leukocytes).
The components of blood
The fluid matrix of blood is known as plasma. Plasma is a straw coloured sticky liquid composed of 90 % water (H2O), and dissolved solids which include proteins (fibrinogen, albumin and globulins), nutrients (glucose, amino acids and lipids), hormones (erythropoietin (EPO) and insulin), and electrolytes such as sodium (Na+) and potassium (K+).
The cells of the blood are the formed elements and perform different functions throughout the body. Let’s take a look at where these cells are made.
The cells of the blood are the formed elements and perform different functions throughout the body. Let’s take a look at where these cells are made.
You may have noticed that many of the cells mentioned in the two videos are immune cells and these are discussed further in the module about the immune system. A large volume (40-45 %) of blood is made-up of red blood cells (RBCs). These cells are extremely important for the transportation of O2.
Erythrocytes - red blood cells are commonly known as RBCs. They are approximately 7.5 micrometres in diameter and are biconcave in shape which enables them to change shape and squeeze through small spaces. They do not contain a nucleus (), nor do they contain other cell organelles such as mitochondria. The lack of nucleus and DNA results in the cells slowly deteriorating and not being able to repair themselves limiting their lifespan to approximately 120 days. Old and damaged RBCs are removed by macrophages in the spleen.
You may now be wondering how RBCs carry out their functions with no mitochondria for the generation of adenosine triphosphate (ATP). They do this through a process known as anaerobic metabolism, meaning without oxygen. Not requiring oxygen makes erythrocytes extremely proficient at their job, which is to carry O2 and deliver this gas to all cells within the body.
Erythrocytes contain a special protein known as haemoglobin (Hb) which takes up most of the cells volume. A haemoglobin molecule contains four iron (Fe) atoms each of which can bind a molecule of O2. O2 diffuses into the blood at the alveoli-capillary interface (in the lungs), where haemoglobin reversibly binds O2 and transports it to all cells. When O2 is bound to haemoglobin it is known as oxyhaemoglobin, which gives blood its bright red colour (and therefore haemoglobin is known as a pigment giving the pink tinge to skin and membranes). At the capillary-cell interface a small amount of CO2 which has diffused across the cell-capillary interface binds to the RBC leading to the unloading of O2 which diffuses across the capillary surface and into the cells. The haemoglobin lacking O2 is known as deoxyhaemoglobin and is found in the veins of the systemic circulation. RBCs can only bind up to 20 % of the CO2 produced as a result of cell activity (metabolism), the remainder is carried to the lungs either dissolved in the blood (~7%) or as bicarbonate ions (HCO3-). This exchange of CO2 with O2 at the capillary bed is known as internal respiration.
Erythrocytes - red blood cells are commonly known as RBCs. They are approximately 7.5 micrometres in diameter and are biconcave in shape which enables them to change shape and squeeze through small spaces. They do not contain a nucleus (), nor do they contain other cell organelles such as mitochondria. The lack of nucleus and DNA results in the cells slowly deteriorating and not being able to repair themselves limiting their lifespan to approximately 120 days. Old and damaged RBCs are removed by macrophages in the spleen.
You may now be wondering how RBCs carry out their functions with no mitochondria for the generation of adenosine triphosphate (ATP). They do this through a process known as anaerobic metabolism, meaning without oxygen. Not requiring oxygen makes erythrocytes extremely proficient at their job, which is to carry O2 and deliver this gas to all cells within the body.
Erythrocytes contain a special protein known as haemoglobin (Hb) which takes up most of the cells volume. A haemoglobin molecule contains four iron (Fe) atoms each of which can bind a molecule of O2. O2 diffuses into the blood at the alveoli-capillary interface (in the lungs), where haemoglobin reversibly binds O2 and transports it to all cells. When O2 is bound to haemoglobin it is known as oxyhaemoglobin, which gives blood its bright red colour (and therefore haemoglobin is known as a pigment giving the pink tinge to skin and membranes). At the capillary-cell interface a small amount of CO2 which has diffused across the cell-capillary interface binds to the RBC leading to the unloading of O2 which diffuses across the capillary surface and into the cells. The haemoglobin lacking O2 is known as deoxyhaemoglobin and is found in the veins of the systemic circulation. RBCs can only bind up to 20 % of the CO2 produced as a result of cell activity (metabolism), the remainder is carried to the lungs either dissolved in the blood (~7%) or as bicarbonate ions (HCO3-). This exchange of CO2 with O2 at the capillary bed is known as internal respiration.
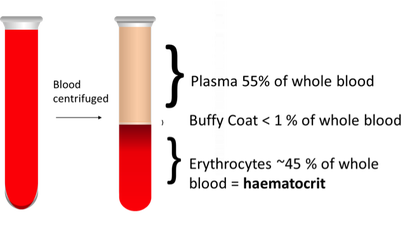
The haematocrit is a measure of the percentage of erythrocytes found in blood. Following centrifugation blood splits into three layers. The top layer is the plasma, the middle layer is the buffy coat and the bottom layer contains the RBCs. The percentage of total blood volume taken-up by RBCs is known as the haematocrit.
It Is important that an individual has enough red blood cells and enough iron to provide sufficient haemoglobin in order to deliver sufficient O2 to all cells in the body. Without oxygen cells cannot perform their necessary functions. Lack of O2 will cause cells to die as it is required for ATP production and an individual with a low number of red blood cells or a lack of haemoglobin will feel tired and listless; they are suffering from anaemia. Blood tests can be performed to check RBC levels (haematocrit) and iron levels.
Now that you understand the importance of the haematocrit you should realise that the numbers of RBCs within the circulation are kept within defined limits, with production being tightly controlled to keep numbers steady. Homeostatic imbalances lead to conditions such as anaemia and tissue hypoxia, or polycythaemia (too many red blood cells) and viscous blood (making the heart work harder to pump against the pressure). Erythrocytes are formed from haematopoietic stem cells in a process known as erythropoiesis. This process is very rapid and is under hormonal control.
Low levels of the hormone erythropoietin (EPO) circulate in blood at all times to maintain a steady state of RBC production. Levels of oxygen are detected by certain cells in the kidneys. When these cells become hypoxic as a result of low RBC count, decreased haemoglobin or low O2 levels the kidney releases erythropoietin which stimulates the red bone marrow to produce more RBCs. This in turn leads to an increase in the number of cells delivering O2 to the kidney. This is one side of a negative feedback homeostatic mechanism.
Low levels of the hormone erythropoietin (EPO) circulate in blood at all times to maintain a steady state of RBC production. Levels of oxygen are detected by certain cells in the kidneys. When these cells become hypoxic as a result of low RBC count, decreased haemoglobin or low O2 levels the kidney releases erythropoietin which stimulates the red bone marrow to produce more RBCs. This in turn leads to an increase in the number of cells delivering O2 to the kidney. This is one side of a negative feedback homeostatic mechanism.
Haemostasis (‘stasis’ means to be still or stop)
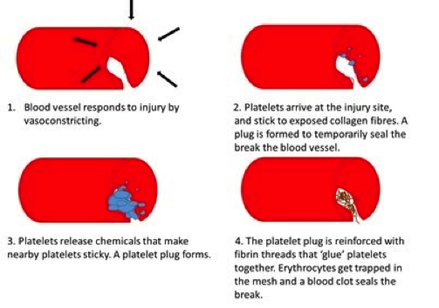
Platelets are cell fragments of megakaryoctyes, and do not contain a nucleus. A hormone called thrombopoietin regulates the production of platelets. Platelets contain granules which enclose chemicals essential for the clotting process. Unless a trauma to a blood vessel occurs, platelets circulate freely through the circulatory system. When a blood vessel is ruptured platelets stick to the site of the injury forming a plug to reduce bleeding. Blood clotting is a positive feedback mechanism ceasing once the clot has successfully stopped the bleeding.
You should now know that:
You should now know that:
- Blood circulates around the body to all cells.
- The contents of blood include formed elements and plasma.
- Erythrocytes carry respiratory gases to and from the lungs to all tissues of the body.
- Erythropoiesis is a negative feedback mechanism.
- When a blood vessel is damaged the activation of platelets is essential to clotting.
- Blood clotting is a positive feedback mechanism.
2 The structure of the heart
The heart is a pump which receives deoxygenated blood from the tissues then pumps it to the lungs to be oxygenated and returns the now oxygenated blood back to the systemic circulation. The heart is about the size of one’s fist and is enclosed within the mediastinum, posterior to the ribs, anterior to the vertebral column, and superior to the diaphragm. It lies obliquely from the second rib to the fifth intercostal space, with about two-thirds of the organ lying to the left of the mid-sternal line. Its apex points inferiorly towards the left hip.
The heart wall consists of epicardium, myocardium and endocardium. The inside layer of the endocardium consists of squamous epithelial cells resting on a layer of connective tissue. This connective tissue lines the heart chambers and covers the fibrous skeleton of the heart valves. The middle layer, the myocardium, consists mainly of cardiac muscle which forms the bulk of the heart. Cardiac muscle tissue is linked together by connective tissue.
Enclosing the heart is a double-walled sac known as the pericardium. The superficial layer is made of fibrous pericardium which protects the heart and anchors it to surrounding structures. This fibrous layer also prevents the heart from overfilling. The serous pericardium lies deep to the fibrous layer and consists of two layers, the parietal layer and the visceral layer (epicardium), which forms part of the heart wall. A cavity exists between the serous and visceral layers containing serous fluid which allows the serous membranes to glide freely past one and other, allowing the heart to beat in a relatively friction-free environment.
The heart wall consists of epicardium, myocardium and endocardium. The inside layer of the endocardium consists of squamous epithelial cells resting on a layer of connective tissue. This connective tissue lines the heart chambers and covers the fibrous skeleton of the heart valves. The middle layer, the myocardium, consists mainly of cardiac muscle which forms the bulk of the heart. Cardiac muscle tissue is linked together by connective tissue.
Enclosing the heart is a double-walled sac known as the pericardium. The superficial layer is made of fibrous pericardium which protects the heart and anchors it to surrounding structures. This fibrous layer also prevents the heart from overfilling. The serous pericardium lies deep to the fibrous layer and consists of two layers, the parietal layer and the visceral layer (epicardium), which forms part of the heart wall. A cavity exists between the serous and visceral layers containing serous fluid which allows the serous membranes to glide freely past one and other, allowing the heart to beat in a relatively friction-free environment.
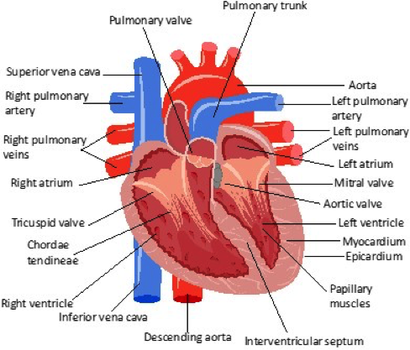
The heart consists of four chambers:
The atria are smaller and thinner walled than the ventricles and receive blood from the systemic circulation and the lungs.
The right ventricle contains less cardiac muscle than the left ventricle and clearly shows how structure relates to function; the right ventricle pumps blood out of the heart to the lungs (only a short distance) via the pulmonary artery, while the left ventricle has to pump blood out of the heart, via the aorta, and around the body to every tissue and cell.
Between the right atria and right ventricle is a valve known as the right atrioventricular or tricuspid valve which allows one-way blood to flow from the right atrium to the right ventricle.
On the left side of the heart the bicuspid or mitral valve lies between the left atrium and the left ventricle, and likewise allows the one-way flow of blood between these two chambers.
Both sets of valves have fine threads known as chordae tendineae attaching them to papillary muscles within the walls of the ventricles. Between the ventricles and their respective arteries are semilunar valves (the pulmonary valve and the aortic valve), which prevent backflow of blood from the arteries.
- two upper chambers known as atrium (plural; atria), and
- two lower chambers known as ventricles.
The atria are smaller and thinner walled than the ventricles and receive blood from the systemic circulation and the lungs.
The right ventricle contains less cardiac muscle than the left ventricle and clearly shows how structure relates to function; the right ventricle pumps blood out of the heart to the lungs (only a short distance) via the pulmonary artery, while the left ventricle has to pump blood out of the heart, via the aorta, and around the body to every tissue and cell.
Between the right atria and right ventricle is a valve known as the right atrioventricular or tricuspid valve which allows one-way blood to flow from the right atrium to the right ventricle.
On the left side of the heart the bicuspid or mitral valve lies between the left atrium and the left ventricle, and likewise allows the one-way flow of blood between these two chambers.
Both sets of valves have fine threads known as chordae tendineae attaching them to papillary muscles within the walls of the ventricles. Between the ventricles and their respective arteries are semilunar valves (the pulmonary valve and the aortic valve), which prevent backflow of blood from the arteries.
Blood flow through the heart
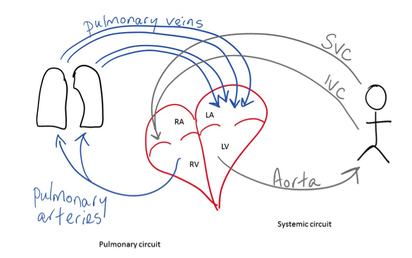
Deoxygenated blood enters the heart via the superior and inferior vena cavae (singular; vena cava), into the right atrium where it passes through the tricuspid valve into the right ventricle. From the right ventricle blood, via the pulmonary semi-lunar valve (pulmonary valve), enters the pulmonary artery and is taken to the lungs where it is oxygenated. This oxygenated blood returns to the left atrium and passes through the bicuspid valve into the left ventricle. From here the blood moves through the aortic semilunar valve (aortic valve), into the aorta, and is transported around the body.
Coronary Arteries
The heart has an absolute requirement for oxygen in order to function, while it can utilise several different nutrition sources which include glucose, fatty acids and lactic acid, it cannot however, utilise oxygen or nutrients from the blood which passes through the heart. The heart therefore, has its own supply of blood vessels known as the coronary circulation. The vessels that supply the heart wall with oxygen are known as the coronary arteries which fill while the ventricles are at rest and those that take away the deoxygenated blood are known as the coronary veins.
Coronary Arteries
The heart has an absolute requirement for oxygen in order to function, while it can utilise several different nutrition sources which include glucose, fatty acids and lactic acid, it cannot however, utilise oxygen or nutrients from the blood which passes through the heart. The heart therefore, has its own supply of blood vessels known as the coronary circulation. The vessels that supply the heart wall with oxygen are known as the coronary arteries which fill while the ventricles are at rest and those that take away the deoxygenated blood are known as the coronary veins.
3 Heart function: pacemaker cells and the conduction of electrical signals
The heart beats without any neural or chemical input. Indeed, one can remove a heart from an organism and it will continue to beat. It is able to do this because it has specialised cells within the endocardium that are auto-rhythmic and form the cardiac conduction system. The cells within this system do not contract but initiate and conduct an electrical signal throughout the myocardium to cause cardiac muscle cells to contract.
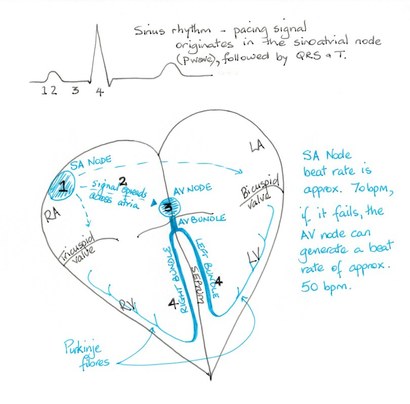
This conduction system consists of (in order of signal direction):
- The Sino-atrial node (SA node), (known as the pacemaker of the heart) is located in the posterior wall of the right atrium.
- The atrioventricular node (AV node), is located in the base of the right atrium between the tricuspid valve and the coronary sinus. There is fibrous tissue surrounding the interventricular valves that insulate the signal in the atria from the ventricular cells. This enables the electrical signal conduction from the AV node into the AV bundle.
- The atrioventricular bundle, (AV bundle, otherwise known as the Bundle of His), which leaves the AV node and passes into the interventricular septum. It follows the path of the septum eventually branching into left and right bundles.These bundle branches descend through the septum towards the apex of the heart. These bundles branches divide further into purkinje fibres which sweep superiorly up through the walls of the ventricles.
The SA node maintains the heartbeat at approximately 70 beats per minute (bpm) and is known as the pacemaker of the heart. If the SA node fails, then the AV node can maintain the heart rate at approximately 50 bpm. The specialised cells of the conduction system have auto-rhythmicity. This means that they can create an electrical signal without any stimulus from the nervous system or from chemical mediators. The SA node is the pacemaker because it can reset more rapidly than other cells in the heart for the next action potential effectively supressing these other cells from setting their own rhythm.
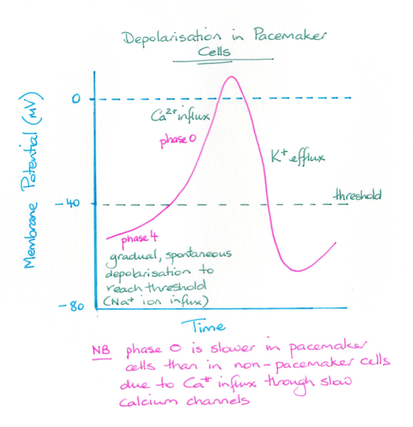
Cells have a membrane potential with the cell being more negative on the inside compared to the outside of the cell. Changes in this membrane potential initiate an electrical signal. In skeletal muscle cells the resting membrane potential was about -70 millivolts (mv). When voltage-gated Na+ channels open Na+ moves into the cell making it more positive. At about -30mv an action potential is created and the cell depolarises. In ventricular cardiac muscle cells the resting membrane potential is around -90mV.
In auto-rhythmic cells there is no resting membrane potential. Na+ channels are always open and once one action potential is complete these cells slowly depolarise to create another action potential. These auto-rhythmic cells have a maximise negative voltage of -60mV. The refractory period of cardiac muscle cells is of much longer duration than those of skeletal cells or nerves. This means that an action potential is generated of about once every 0.8 sec.
In auto-rhythmic cells there is no resting membrane potential. Na+ channels are always open and once one action potential is complete these cells slowly depolarise to create another action potential. These auto-rhythmic cells have a maximise negative voltage of -60mV. The refractory period of cardiac muscle cells is of much longer duration than those of skeletal cells or nerves. This means that an action potential is generated of about once every 0.8 sec.
4 The cardiac muscle and how it contracts
As the cardiac muscle has an electrical signal, it is possible for it to convert this impulse into a contraction. As was stated previously the electrical signal occurs approximately once every 0.8 sec, meaning that in an average individual the heart muscle will receive 75 impulses every minute and hence will contract 75 time in one min. For those of you mathematically minded this is simply;
60 sec /0.8 sec /beat = 75 bpm
So how does this muscle contraction come about.
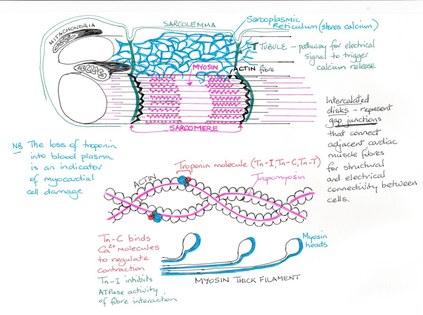
The cardiac muscle (myocardium) is similar to skeletal muscle in that it is striated and contracts by the sliding filament mechanism. The cells are however, relatively short, branch and are interconnected. Each muscle fibre contains one or sometimes two nuclei and the intercellular spaces are filled with endomysium. Unlike skeletal muscle fibres which are independent of one and other, cardiac cells contain intercalated discs which consist of desmosomes and gap junctions. Desmosomes are proteins which prevent cardiac muscle from pulling apart. Gap junctions are protein pores which lie between the sarcolemma of adjacent cardiac cells and allow the movement of ions between these cells thus allowing an action potential to travel throughout the cardiomyocytes so that they contract as one. This contracting of the muscle as a single unit is known as a functional syncytium and is important in that the heart must contract as one if it is to be an effective pump. Mitochondria are numerous, approximately 25-35% of the volume of these cells making them resistant to fatigue. The remaining volume of the cell is made up of myofibrils composed of sarcomeres containing Z-discs. T-tubules are present but are fewer and wider than in skeletal muscle and only enter the cell once per sarcomere, at the Z-disc.
You have already discovered that some cardiac muscle cells are self-excitable and they initiate the depolarisation of the entire cardiac muscle. This wave of depolarisation spreads throughout the whole muscle via the conduction system and gap junctions. Depolarisation starts with a few fast voltage-gated Na+ channels opening in the sarcolemma allowing Na+ to flow into the cell and depolarising the cell from -90 mv to +30 mv. at which time the Na+ channels close. Depolarisation triggers the opening of K+ channels and K+ exits the cardiomyocytes. There is a small decrease in the membrane potential which cause Ca2+ channels in the sarcolemma to open and Ca2+ enters the interstitial fluid, stimulates the sarcoplasmic reticulum to open of more CA2+ channels. This movement of K+ out and Ca2+ in to the sarcoplasmic reticulum means that there is no change in the membrane potential it remains in a depolarised state known as the plateau phase. Ca2+ channels eventually close and K+ continues to move out of the cardiomyocytes, repolarising the cells.
The movement of Ca2+ into the sarcoplasm initiates the events of cardiac muscle contraction; the binding of Ca2+ to troponin and cross-bridge cycling within the sarcomere, in a similar way to that of skeletal muscle. It should be noted however, that cardiac muscle cannot sustain muscle contraction without relaxation.
60 sec /0.8 sec /beat = 75 bpm
So how does this muscle contraction come about.
The cardiac muscle (myocardium) is similar to skeletal muscle in that it is striated and contracts by the sliding filament mechanism. The cells are however, relatively short, branch and are interconnected. Each muscle fibre contains one or sometimes two nuclei and the intercellular spaces are filled with endomysium. Unlike skeletal muscle fibres which are independent of one and other, cardiac cells contain intercalated discs which consist of desmosomes and gap junctions. Desmosomes are proteins which prevent cardiac muscle from pulling apart. Gap junctions are protein pores which lie between the sarcolemma of adjacent cardiac cells and allow the movement of ions between these cells thus allowing an action potential to travel throughout the cardiomyocytes so that they contract as one. This contracting of the muscle as a single unit is known as a functional syncytium and is important in that the heart must contract as one if it is to be an effective pump. Mitochondria are numerous, approximately 25-35% of the volume of these cells making them resistant to fatigue. The remaining volume of the cell is made up of myofibrils composed of sarcomeres containing Z-discs. T-tubules are present but are fewer and wider than in skeletal muscle and only enter the cell once per sarcomere, at the Z-disc.
You have already discovered that some cardiac muscle cells are self-excitable and they initiate the depolarisation of the entire cardiac muscle. This wave of depolarisation spreads throughout the whole muscle via the conduction system and gap junctions. Depolarisation starts with a few fast voltage-gated Na+ channels opening in the sarcolemma allowing Na+ to flow into the cell and depolarising the cell from -90 mv to +30 mv. at which time the Na+ channels close. Depolarisation triggers the opening of K+ channels and K+ exits the cardiomyocytes. There is a small decrease in the membrane potential which cause Ca2+ channels in the sarcolemma to open and Ca2+ enters the interstitial fluid, stimulates the sarcoplasmic reticulum to open of more CA2+ channels. This movement of K+ out and Ca2+ in to the sarcoplasmic reticulum means that there is no change in the membrane potential it remains in a depolarised state known as the plateau phase. Ca2+ channels eventually close and K+ continues to move out of the cardiomyocytes, repolarising the cells.
The movement of Ca2+ into the sarcoplasm initiates the events of cardiac muscle contraction; the binding of Ca2+ to troponin and cross-bridge cycling within the sarcomere, in a similar way to that of skeletal muscle. It should be noted however, that cardiac muscle cannot sustain muscle contraction without relaxation.
5 The normal ECG and what it represents
The ECG is a method of measuring electrical signals within the heart using electrodes attached to the skin of the ankles, wrists and the chest. A recording is made which can be read like a graph.
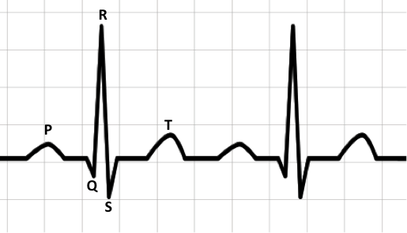
A normal ECG consists of P, QRS and T waves that represent a sinus rhythm, that is the rhythm commences in the sinus node of the conduction system.
Atrial repolaristion occurs during ventricular depolarisation, and is a smaller upward deflection as a result of the differences in the muscle size between atria and ventricles, hence the wave representing atrial repolarisation cannot be seen on the ECG.
A normal ECG consists of P, QRS and T waves that represent a sinus rhythm, that is the rhythm commences in the sinus node of the conduction system.
- The P wave represents atrial depolarisation leading to atrial contraction. The P wave originates in the SA node (the pacemaker) and lasts between 0.08 and 1 second.
- The QRS complex represents ventricular depolarisation which results in ventricle contraction. The QRS lasts between 0.06 and 1 second.
- The T wave represents ventricular repolaristion at the end of which the resting membrane potential is regained.
Atrial repolaristion occurs during ventricular depolarisation, and is a smaller upward deflection as a result of the differences in the muscle size between atria and ventricles, hence the wave representing atrial repolarisation cannot be seen on the ECG.
A three lead ECG gives an ECG trace that indicates the electrical signal as it moves across the anterior surface of the heart as depicted in the drawing. A 12 lead ECG provides traces from different heart surfaces moving from anterior to lateral. However, all these traces should still display the three waves: P, QRS and T.12 lead traces are useful for determining where in the heart a conduction may be faulty such as during a heart attack or for those with conduction system defects.
The Cardiac Cycle
The cardiac cycle describes the events that occur between the start of one heart beat and the start of the next.
During this period the heart chambers contract (systole) and relax (diastole). When the heart muscle contracts there is an increase in pressure within the heart chamber and this forces blood out of that chamber, when the heart muscle relaxes the pressure inside the chamber decreases and blood can move into that chamber.
The alternating contraction and relaxation of both the atria and the ventricles forces blood unidirectionally, with the heart valves opening and closing to prevent the back flow of blood.
During this period the heart chambers contract (systole) and relax (diastole). When the heart muscle contracts there is an increase in pressure within the heart chamber and this forces blood out of that chamber, when the heart muscle relaxes the pressure inside the chamber decreases and blood can move into that chamber.
The alternating contraction and relaxation of both the atria and the ventricles forces blood unidirectionally, with the heart valves opening and closing to prevent the back flow of blood.
6 The structure of different types of blood vessels
Blood vessels are the conduits that carry blood from the heart to the extremities of the body, delivering oxygen and nutrients, and then return the blood containing carbon dioxide and waste back to the heart. Blood vessels form the circulatory system and are a continuous length of tubes varying in diameter from 8 mm the (smallest capillary) to 25 mm in diameter (the largest artery).
Blood flow through the capillaries
Capillaries are the smallest blood vessels in the body, approximately 1 mm in length and 8-10 micrometres in diameter. They have thin endothelial walls which are stabilised by smooth muscle-like cells known as pericytes. These pericytes also help to control the permeability of the capillary.
Most tissues have a rich network of capillaries which gives direct access for blood to nearly every cell in the body. There are three types of capillaries:
Most tissues have a rich network of capillaries which gives direct access for blood to nearly every cell in the body. There are three types of capillaries:
- Continuous capillaries are the most common and are abundant in skin and muscle tissue. Their endothelial cells contain tight junctions which makes the lining of these vessel continuous. Along this lining are intercellular clefts which allow for the passage of fluids and solutes.
- Fenestrated capillaries contain endothelial cells which are filled with pores making them ideal for active absorption and filtration of fluids and solutes. Knowing this function, where do you think you might find this type of capillary?
- Sinusoid capillaries have large irregularly shaped lumens and are fenestrated, allowing the passage of large molecules between the blood and the tissues. These capillaries are only found in the liver, bone marrow, spleen and adrenal medulla.
Capillary beds
Capillaries act together forming a network of interweaving vessels through which blood runs from the terminal arteriole to the post-capillary venule. This is the capillary bed and forms the microcirculation where the exchange of gases and nutrients/waste takes place between the blood and the tissues.
True capillaries branch-off a metarteriole which forms a continuous thoroughfare channel from the arteriole end to the venule end of the capillary bed. True capillaries number between 10 – 100, depending on the organ they serve and are the vessels through which exchange takes place.
Blood flow through the capillary bed is controlled by precapillary sphincters, a band of smooth muscle, that surround the true capillaries at the point where they branch-off the metarteriole. Depending on oxygen and nutrient requirements of the tissue, these sphincters may relax allowing blood to flow into the capillaries and participate in exchanges, or constrict, allowing blood to bypass the tissue.
O, CO, nutrients and waste products pass between the blood and interstitial fluid by diffusion, that is down their concentration gradient.
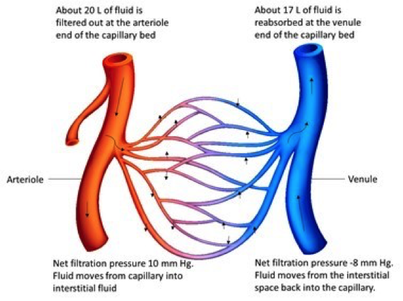
At the same time, bulk fluid flow moves fluid out of the blood at the arteriole end of the capillary bed and returns most of this fluid at the venous end. This bulk flow is important for maintaining blood and interstitial fluid volumes. The direction and amount of bulk flow is determined by two opposing forces; hydrostatic pressure and colloid osmotic pressure.
True capillaries branch-off a metarteriole which forms a continuous thoroughfare channel from the arteriole end to the venule end of the capillary bed. True capillaries number between 10 – 100, depending on the organ they serve and are the vessels through which exchange takes place.
Blood flow through the capillary bed is controlled by precapillary sphincters, a band of smooth muscle, that surround the true capillaries at the point where they branch-off the metarteriole. Depending on oxygen and nutrient requirements of the tissue, these sphincters may relax allowing blood to flow into the capillaries and participate in exchanges, or constrict, allowing blood to bypass the tissue.
O, CO, nutrients and waste products pass between the blood and interstitial fluid by diffusion, that is down their concentration gradient.
At the same time, bulk fluid flow moves fluid out of the blood at the arteriole end of the capillary bed and returns most of this fluid at the venous end. This bulk flow is important for maintaining blood and interstitial fluid volumes. The direction and amount of bulk flow is determined by two opposing forces; hydrostatic pressure and colloid osmotic pressure.
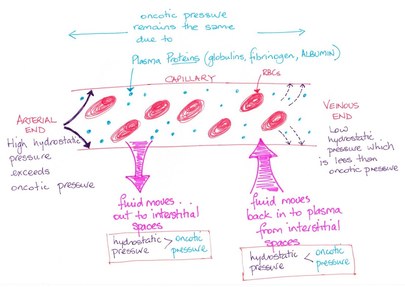
Hydrostatic pressure (HPC) is the pressure exerted by the blood on the capillary walls, (this is the same as blood pressure in the capillaries), and forces fluid out of the capillaries by filtration.
Blood cells and proteins cannot cross the capillary bed and remain in the blood.
As blood flows through the capillary bed blood pressure drops and thus hydrostatic pressure decreases from 35 mm Hg to 17 mm Hg. This HPC is opposed by HP in the interstitial fluid (HPIF).
There is however, very little fluid in the interstitial space and so the amount of HPIF is negligible. Remember I said there is a force opposing HP. This force is known as colloid osmotic pressure (OP). This pressure is caused by molecules that are too large to cross the capillary wall, and in the capillary fluid is pulled towards these molecules, that is back into the blood.
Capillary colloid OP (OPC) Interstitial fluid contains few proteins, relative to the blood, and has an interstitial (OPIF) of approximately 0.5 - 1 mm Hg and again does not vary greatly from the arteriole to the venule end of the capillary bed.
Blood cells and proteins cannot cross the capillary bed and remain in the blood.
As blood flows through the capillary bed blood pressure drops and thus hydrostatic pressure decreases from 35 mm Hg to 17 mm Hg. This HPC is opposed by HP in the interstitial fluid (HPIF).
There is however, very little fluid in the interstitial space and so the amount of HPIF is negligible. Remember I said there is a force opposing HP. This force is known as colloid osmotic pressure (OP). This pressure is caused by molecules that are too large to cross the capillary wall, and in the capillary fluid is pulled towards these molecules, that is back into the blood.
Capillary colloid OP (OPC) Interstitial fluid contains few proteins, relative to the blood, and has an interstitial (OPIF) of approximately 0.5 - 1 mm Hg and again does not vary greatly from the arteriole to the venule end of the capillary bed.
So, what does all this mean?
The hydrostatic and colloid osmotic pressures determine which way fluid will move across the capillary wall. That is the net filtration pressure (NFP).
At the arteriole end of the capillary bed:
NFP = (HPC + OPIF) - (HPIF + OPC)
(35 + 1) – (0 + 26) = 10 mm Hg (net outward pressure).
This means that the drive to push fluid out of the capillary is greater than that to pull it back in. Fluid moves from the capillary into the interstitial space.
At the venule end of the capillary bed:
NFP = (HPC + OPIF) - (HPIF + OPC)
(17 + 1) – (0 + 26) = -8 mm Hg (net outward pressure).
This means that the drive to reabsorb fluid into the capillary is greater than that to push it out. Fluid moves from the interstitial space into the capillary.
The hydrostatic and colloid osmotic pressures determine which way fluid will move across the capillary wall. That is the net filtration pressure (NFP).
At the arteriole end of the capillary bed:
NFP = (HPC + OPIF) - (HPIF + OPC)
(35 + 1) – (0 + 26) = 10 mm Hg (net outward pressure).
This means that the drive to push fluid out of the capillary is greater than that to pull it back in. Fluid moves from the capillary into the interstitial space.
At the venule end of the capillary bed:
NFP = (HPC + OPIF) - (HPIF + OPC)
(17 + 1) – (0 + 26) = -8 mm Hg (net outward pressure).
This means that the drive to reabsorb fluid into the capillary is greater than that to push it out. Fluid moves from the interstitial space into the capillary.
7 Cardiovascular haemodynamics
Haemodynamics refers to the cardiac physiology dealing with the forces generated by the heart as it pumps blood.
Remember that the cardiac output in mLs/min is the result of the heart pump rate per minute multiplied by the stroke volume (the volume of blood in millilitres (mLs) pumped with each contraction): CO = HR x SV
We need to look at the factors that impact on this relationship.
Remember that the cardiac output in mLs/min is the result of the heart pump rate per minute multiplied by the stroke volume (the volume of blood in millilitres (mLs) pumped with each contraction): CO = HR x SV
We need to look at the factors that impact on this relationship.
Blood Flow, Blood Pressure and Resistance
The volume of blood flowing through a blood vessel, an organ or the entire circulation is known as the blood flow (F).
It is measured in ml/min and is equivalent to cardiac output.
Blood pressure (BP) is the force that blood exerts on the vessel wall and is measure in millimetres of mercury (mmHg).
Resistance (R) is the friction the blood encounters as it moves through the blood vessels and is affected by blood viscosity, vessel diameter and vessel length.
Blood flow is related to the change in BP and R. This means that if we increase pressure and do not change resistance blood flow will increase, or if we increase resistance but not change the pressure then blood flow will decrease. Resistance has the greatest impact on blood flow because it changes with alteration in blood vessel diameter; during exercise blood vessels serving skeletal muscle will dilate, decreasing resistance and thus increasing blood flow to the muscles.
It is measured in ml/min and is equivalent to cardiac output.
Blood pressure (BP) is the force that blood exerts on the vessel wall and is measure in millimetres of mercury (mmHg).
Resistance (R) is the friction the blood encounters as it moves through the blood vessels and is affected by blood viscosity, vessel diameter and vessel length.
Blood flow is related to the change in BP and R. This means that if we increase pressure and do not change resistance blood flow will increase, or if we increase resistance but not change the pressure then blood flow will decrease. Resistance has the greatest impact on blood flow because it changes with alteration in blood vessel diameter; during exercise blood vessels serving skeletal muscle will dilate, decreasing resistance and thus increasing blood flow to the muscles.
Systemic blood pressure
Blood pressure is result of blood flow opposed by resistance.
The pumping action of the heart creates the flow and the vessels create the resistance.
Blood pressure is highest when blood leaves the heart and enters the aorta, and decreases throughout the systemic circulation reaching 0 mmHg on return to the right atrium.
Two factors affect BP:
When the left ventricle contracts the blood forced into the aorta distending it. This pressure is 120 mmHg and this is known as the systolic pressure. As the ventricle relaxes, the elastic fibres of the walls of the aorta enable it to recoiland the pressure in the vessel drops to about 80 mmHg. This is the diastolic pressure.
The difference between systolic and diastolic pressure is known as pulse pressure. If the systolic pressure is 120mmHg and the diastolic pressure is 80mmHg then the pulse pressure is 40mmHg. Pulse pressure: 120 – 80 = 40 mmHg.
The aortic pressure fluctuates over time. An important measurement is the mean arterial pressure (MAP), which is the pressure which pushes blood to the tissues.
MAP = diastolic pressure + ( pulse pressure / 3)
Both pulse pressure and MAP decline as blood moves further from the heart.
The pumping action of the heart creates the flow and the vessels create the resistance.
Blood pressure is highest when blood leaves the heart and enters the aorta, and decreases throughout the systemic circulation reaching 0 mmHg on return to the right atrium.
Two factors affect BP:
- the compliance (distensibility) of the arteries, and
- the amount of blood forced into the vessels, which changes with activity level.
When the left ventricle contracts the blood forced into the aorta distending it. This pressure is 120 mmHg and this is known as the systolic pressure. As the ventricle relaxes, the elastic fibres of the walls of the aorta enable it to recoiland the pressure in the vessel drops to about 80 mmHg. This is the diastolic pressure.
The difference between systolic and diastolic pressure is known as pulse pressure. If the systolic pressure is 120mmHg and the diastolic pressure is 80mmHg then the pulse pressure is 40mmHg. Pulse pressure: 120 – 80 = 40 mmHg.
The aortic pressure fluctuates over time. An important measurement is the mean arterial pressure (MAP), which is the pressure which pushes blood to the tissues.
MAP = diastolic pressure + ( pulse pressure / 3)
Both pulse pressure and MAP decline as blood moves further from the heart.
Effects of sympathetic and parasympathetic stimulation on cardiac muscle cells and heart rate
Blood pressure is controlled physiologically in several ways.
Neural controls alter both cardiac output and peripheral resistance to maintain MAP by altering blood vessel diameter and blood distribution, and involve the baroreceptors and chemoreceptors.
Neural controls alter both cardiac output and peripheral resistance to maintain MAP by altering blood vessel diameter and blood distribution, and involve the baroreceptors and chemoreceptors.
Baroreceptors are pressure sensors found in the aorta and the internal carotid arteries.
When heart rate and CO increase, they detect the resulting rise in BP and signal to the medulla oblongata which in turn increases vagal tone (parasympathetic system) resulting in vasodilation of the arterioles, venodilation and slowing of the heart rate, which then decreases CO.
Conversely, if there is a drop in BP then sympathetic output is increased to raise the heart rate and vessel tone to bring CO and BP back to normal.
Chemoreceptors, found in the aortic arch, the carotid arteries, respond to increased CO2 levels, decreased pH and decreased O2.
When heart rate and CO increase, they detect the resulting rise in BP and signal to the medulla oblongata which in turn increases vagal tone (parasympathetic system) resulting in vasodilation of the arterioles, venodilation and slowing of the heart rate, which then decreases CO.
Conversely, if there is a drop in BP then sympathetic output is increased to raise the heart rate and vessel tone to bring CO and BP back to normal.
Chemoreceptors, found in the aortic arch, the carotid arteries, respond to increased CO2 levels, decreased pH and decreased O2.
Hormonal controls involved in BP maintenance
Several hormones are involved in controlling BP:
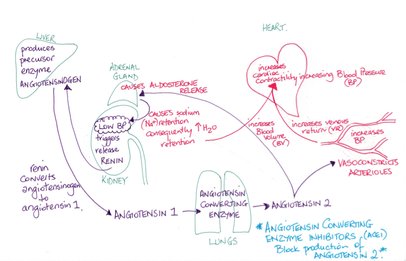
Angiotensin II
The synthesis of angiotensin II is controlled by the nervous system and leads to the release of other hormones. The liver produces and continuously releases angiotensinogen which circulates in the blood. If stimulated by the sympathetic nervous system or if BP falls then the kidneys produce renin which in turn converts angiotensinogen to angiotensin I. Another enzyme angiotensin-converting enzyme (ACE) converts angiotensin I to angiotensin II. Angiotensin II cause vasoconstriction, and regulates blood volume by acting on the kidneys to reduce the formation of urine. Both these actions lead to an increase in BP.
Aldosterone
Aldosterone is released from the adrenal cortex in response to angiotensin II. It increases the reabsorption of Na+ in the kidneys which in turn leads to the reabsorption of water, thus increasing blood volume and therefore BP.
Anti-diuretic hormone (ADH)
The hypothalamus (in the brain), detects increased osmolarity of the blood, usually due to low blood volume and thus low BP, or is stimulated by angiotensin II. It sends nerve signals to the posterior pituitary release ADH. ADH acts on the kidneys to reduce the loss of urine by increasing water reabsorption. ADH also stimulates the thirst centre in the hypothalamus leading to increased fluid intake and thus increased blood volume. Increased blood volume leads to increased BP.
In the case of severe fluid loss, for example during haemorrhaging, then large amounts of ADH are produced, this leads to vasoconstriction and this results in increased peripheral resistance and increased BP.
Atrial Natriuretic Peptide (ANP)
ANP is released by the right atrium of the heart in response to increased venous return which causes the atrial wall to stretch. ANP stimulates vasodilation and increase urine output, resulting in lowered peripheral resistance and lowered blood volume. The nett effect is that BP is lowered.
Angiotensin II
The synthesis of angiotensin II is controlled by the nervous system and leads to the release of other hormones. The liver produces and continuously releases angiotensinogen which circulates in the blood. If stimulated by the sympathetic nervous system or if BP falls then the kidneys produce renin which in turn converts angiotensinogen to angiotensin I. Another enzyme angiotensin-converting enzyme (ACE) converts angiotensin I to angiotensin II. Angiotensin II cause vasoconstriction, and regulates blood volume by acting on the kidneys to reduce the formation of urine. Both these actions lead to an increase in BP.
Aldosterone
Aldosterone is released from the adrenal cortex in response to angiotensin II. It increases the reabsorption of Na+ in the kidneys which in turn leads to the reabsorption of water, thus increasing blood volume and therefore BP.
Anti-diuretic hormone (ADH)
The hypothalamus (in the brain), detects increased osmolarity of the blood, usually due to low blood volume and thus low BP, or is stimulated by angiotensin II. It sends nerve signals to the posterior pituitary release ADH. ADH acts on the kidneys to reduce the loss of urine by increasing water reabsorption. ADH also stimulates the thirst centre in the hypothalamus leading to increased fluid intake and thus increased blood volume. Increased blood volume leads to increased BP.
In the case of severe fluid loss, for example during haemorrhaging, then large amounts of ADH are produced, this leads to vasoconstriction and this results in increased peripheral resistance and increased BP.
Atrial Natriuretic Peptide (ANP)
ANP is released by the right atrium of the heart in response to increased venous return which causes the atrial wall to stretch. ANP stimulates vasodilation and increase urine output, resulting in lowered peripheral resistance and lowered blood volume. The nett effect is that BP is lowered.
Factors effecting the force of contraction
The pressure of blood returning to the ventricles will influence the force of the ventricular contraction. Venous return describes the blood flow back to the heart from the systemic circulation and determines the ventricular preload pressure (the pressure created by the amount of blood that has filled the ventricle). Venous return will increase when blood pressure increases. The higher the preload the more forceful the ventricular contraction and more blood is forced out of the chamber increasing the stroke volume. This ultimately increases the blood pressure.
As an example – when you begin to run your leg muscles work harder pushing more blood back to heart. This increases the venous return and the volume of blood in the chambers; the preload. The extra stretch created in the ventricular muscle will cause it to contract with more force, emptying more blood from the chamber. At the same time the muscle tissues need more oxygen to achieve an increased workload as you run and this will cause the heart to respond by increasing the heart rate. Since cardiac output is a product of stroke volume and heart rate, the cardiac output is raised increasing the blood pressure. This positive feedback loop will be maintained until you cease running, keeping the blood pressure at a level necessary to meet needs of the cells.
Afterload is the pressure in the aorta created by the fact that there is always blood filling the vessels and so the heart must pump hard enough to move fluid into the aorta against that pressure. When peripheral resistance is increased, the heart must pump with more force to overcome that pressure. In people with vessel disease such as atherosclerosis (lipid deposits in vessel walls that decrease the vessels elasticity and capacity to vasodilate or constrict) then the peripheral resistance is increased. Their heart has to pump harder to overcome this resistance leading to high blood pressure.
As an example – when you begin to run your leg muscles work harder pushing more blood back to heart. This increases the venous return and the volume of blood in the chambers; the preload. The extra stretch created in the ventricular muscle will cause it to contract with more force, emptying more blood from the chamber. At the same time the muscle tissues need more oxygen to achieve an increased workload as you run and this will cause the heart to respond by increasing the heart rate. Since cardiac output is a product of stroke volume and heart rate, the cardiac output is raised increasing the blood pressure. This positive feedback loop will be maintained until you cease running, keeping the blood pressure at a level necessary to meet needs of the cells.
Afterload is the pressure in the aorta created by the fact that there is always blood filling the vessels and so the heart must pump hard enough to move fluid into the aorta against that pressure. When peripheral resistance is increased, the heart must pump with more force to overcome that pressure. In people with vessel disease such as atherosclerosis (lipid deposits in vessel walls that decrease the vessels elasticity and capacity to vasodilate or constrict) then the peripheral resistance is increased. Their heart has to pump harder to overcome this resistance leading to high blood pressure.
Bibliography
Marieb, N. M. & Hoehn, K. (2013). Human Anatomy and Physiology. (9th Edition). Person International Edition. USA:Pearson Education Inc.
Saladin, K. S. (2015). Anatomy and Physiology. The Unity of Form and Function. (7th Edition). USA:McGraw-Hill Education.
Saladin, K. S. (2015). Anatomy and Physiology. The Unity of Form and Function. (7th Edition). USA:McGraw-Hill Education.